The largest microbial community in the body is found in the gut, where trillions of microbes reside. These microorganisms help break down food, produce essential vitamins, regulate immune function, and even influence our mood.2 For more information on what the microbiome is, check out our post on The Amazing Gut Microbiome: What It Is and Why It Matters.
Just as no two people are exactly alike, neither are their gut microbiomes. In fact, the microbiome is unique enough to feasibly identify us, like a fingerprint, shown by researchers at Harvard. Dysbiosis is a general term that refers to a disruption to the microbiome that results in an imbalance of the proportions of microbes that usually exist, usually from overgrowth of a harmful organism or undergrowth of a beneficial organism. Research has identified associations between certain dysbiotic microbial patterns and various symptoms and conditions, so understanding your unique microbiome composition can improve your understanding of your health. We can think of these personalized insights like a customized roadmap for your set of symptoms. From unexplained fatigue to digestive discomfort, these insights can help tailor solutions to address your individual needs, potentially providing answers and a path to better health.
1. Digestive Issues
Digestive issues are one of the most common signs of an unhealthy gut. Microbiome profiles of individuals with digestive issues have been observed to exhibit differences in microbial composition. For example, researchers have observed increased Blautia species and increased Faecalibacteria as markers of a ‘healthy’ gut. A healthy gut microbiome is further characterized by beneficial commensal bacteria like Bifidobacterium and Lactobacillus, while an unhealthy gut experiencing digestive issues may have increased levels of Clostridium difficile, Escherichia coli and Helicobater pylori which have the potential to cause infection and inflammation in the gut. Such changes in the gut microbiome have been correlated with altered gut motility, which can lead to uncomfortable symptoms such as bloating, gas, diarrhea, or constipation.
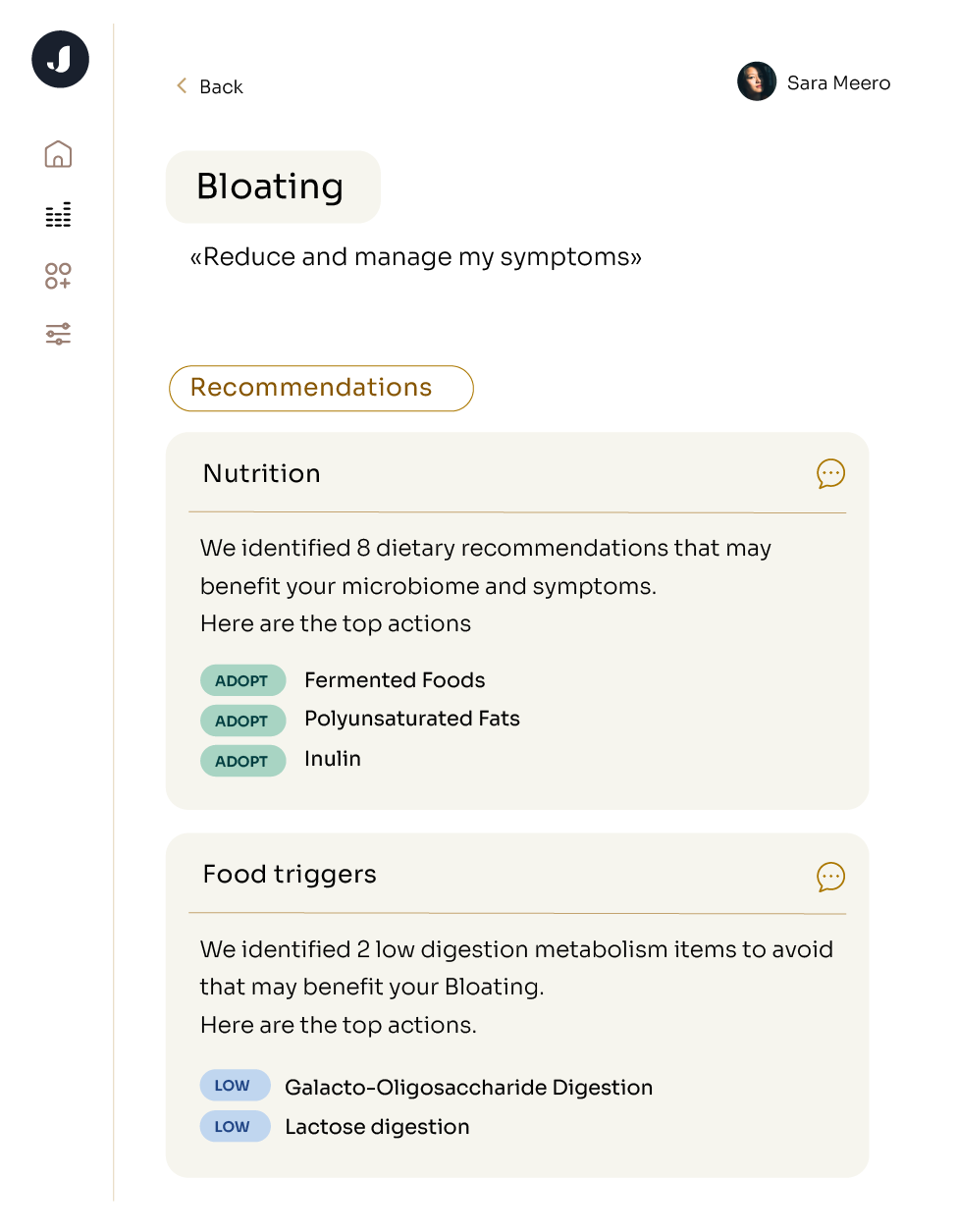
Bloating
Bloating is a common symptom of an unhealthy gut and occurs when your stomach feels swollen or distended after eating. This can be caused by excessive gas production within the gut and build up, causing discomfort and bloating. In a study looking at otherwise healthy people, levels of Anaerotruncus colihominis, Ruminococcus callidus, and Lachnospira pectinoschiza and related species increased more than tenfold during bloating episodes. People with irritable bowel syndrome (IBS) often experience these bloating episodes, and it's been found that Ruminococcaceae are associated with these episodes in IBS. However, it remains uncertain whether this connection is a cause of the symptoms by causing fermentation and gas production in the gut or a result of IBS itself.
Diarrhea & Constipation
Several factors can trigger diarrhea, including infections, antibiotics or changes in diet. In cases of infection, the introduction of harmful pathogens can disrupt the microbiome, leading to an overgrowth of certain bacteria, such as Shigella or Clostridium difficile, or a decrease in beneficial ones, such as Lactobacillus. Constipation can also be caused by changes in diet, dehydration, lack of physical activity, pain medications or various medical conditions. Of note, a lack of dietary fiber can lead to constipation. People with constipation can have variations in their gut bacteria influenced by diet and the speed of food passage through the colon. However, researchers discovered that, even considering these factors, those with constipation often show a higher presence of Bacteroidetes in their colons.
2. Food Intolerances
The gut microbiome may play a role in the development of food sensitivities and food allergies in infancy, as researchers have observed differences in the gut microbiomes of infants and children with food allergy compared to healthy controls. The term allergic sensitization refers to the initial triggering of an allergic immune response when exposed to an allergen. This means that when you encounter a substance to which you are allergic for the first time, your immune system starts to react. Studies have reported that observed altered abundances of Clostridiales, Bacteroidales, Enterobacteriales, and Lactobacillales are associated with sensitization to certain foods.
It has been proposed that increased intestinal permeability, also known as leaky gut syndrome, may also play a role in the development of food allergy. This syndrome has been hypothesized to allow undigested food particles and toxins to leak into the bloodstream, triggering an immune response. Over time, this chronic immune response could contribute towards the development of allergies and food sensitivities.
Common food sensitivities that have been associated with microbiome imbalances include gluten, dairy, soy, and certain types of FODMAPs (fermentable oligosaccharides, disaccharides, monosaccharides, and polyols). These sensitivities can vary from person to person, and it's important to identify the specific triggers for your own gut health.⁵
3. Nutrient Deficiencies
Adequate nutrient absorption is a lesser known function of the gut microbiome. Most nutrient absorption and digestion occur in the stomach and small intestine, which break down and absorb 85% of carbohydrates and 65-90% of proteins. The remaining nutrients end up in your large intestine, called the colon, where microbes allow for further nutrient absorption. This represents 10-30% of total ingested energy, so these microbes allow us to utilize a significant amount of additional energy. Without the activity of the microbiome, from bacteria such as Bacteroides thetaiotaomicron, the colon has limited digestive capacity. These microbes also produce vitamins. Studies report that Firmicutes, Proteobacteria, Bacteroidetes and Actinobacteria are bacteria partly responsible for vitamin synthesis in the gut. A dysbiotic gut microbiome may lead to a reduction in the microbes that are responsible for the breakdown and absorption of nutrients from food which may prevent your body from extracting the full benefits of your food. This interference could mean deficiencies in essential vitamins, minerals, and other nutrients that are necessary for optimal health and wellbeing, though it’s important to note that vitamin deficiencies can be complex and caused by other disorders.
Vitamin and mineral deficiencies can have a wide range of effects on your body. For example, a deficiency in vitamin B12 can cause fatigue, weakness, and even nerve damage. Low levels of iron can lead to anemia, resulting in symptoms such as fatigue, dizziness, and shortness of breath. Deficiencies in vitamins A and C can weaken your immune system and make you more susceptible to infections.
4. Skin Conditions
Signs that you are experiencing skin problems may be found in the gut, especially considering that many skin conditions are systemic and affect more than one part of the body. For example, psoriasis is characterized by systemic inflammation and is accompanied by lower numbers of Akkermansia muciniphila and Faecalibacterium prausnitzii in the gut. Both these bacteria are short-chain fatty acid producers in the gut, which protects against inflammation in the gut. There seems to be a gut-skin link, as evidenced by observations such as the potential prevention of atopic dermatitis (AD) with probiotics.
Additionally, the nutrient deficiencies that we discussed previously can also impact your skin health. For instance, a lack of omega-3 fatty acids can contribute to dry and inflamed skin. Deficiencies in zinc and vitamin E can impair wound healing and increase the risk of skin infections.
5. Mood Disorders
It may surprise you to learn that the gut and the brain are closely connected. In fact, over 95% of total body serotonin is produced in your gut. Many other neurotransmitters such as GABA, catecholamines, and tryptophan are produced in the gut, as well. The gut has its own nervous system, known as the enteric nervous system, which communicates with the brain through the gut-brain axis. There is evidence that the gut microbiome can also influence the brain through immune and hormonal pathways, while changes in the brain can influence the composition of the gut microbiome.
Studies have observed that individuals with mood disorders have gut microbiomes characterized by alterations in the abundance of certain bacteria in the gut, such as decreased bacteria that generate short-chain fatty acids and increased pro-inflammatory bacteria. The abundances of Actinobacteria and Enterobacteriaceae were increased and Faecalibacterium was decreased consistently in patients with either bipolar disorder or major depressive disorder. Similarly, bacteria that produce short-chain fatty acids such as Coprococcus have been associated to be found in the guts of patients reporting greater reported emotional well being while low levels found in people with depression.¹²
6. Autoimmune Disorders
Autoimmune disorders occur when the immune system mistakenly attacks and damages the body's own tissues, organs, and cells. Some autoimmune conditions are well known, for example type 1 diabetes, celiac disease, Hashimoto’s disease and rheumatoid arthritis, while others are lesser known and more difficult to diagnose. These conditions are believed to be caused by both genetic predisposition and environmental triggers, such as diet and lifestyle, though the exact development of these disorders remain unclear.
People who have autoimmune disease experience systemic inflammation which affects multiple organs and parts of their bodies, and this inflammation may be associated with certain patterns of microbes in their microbiome. For example, lupus has been associated with a lower Firmicutes/Bacteroidetes ratio, a decrease in the abundance of Lactobacillaeceae and an increase in Lachnospiraceae. An increased amount of Firmicutes has been observed in both rheumatoid arthritis and Sjogren’s syndrome. In comparison to healthy subjects, in multiple sclerosis, Prevotella decreases, while in rheumatoid arthritis, Prevotella increases.,, There is also some evidence to suggest that administration of probiotics, which help change the composition of the gut microbiome, can potentially alleviate symptoms of some autoimmune diseases in specific circumstances, yet evidence in this space is still emerging.
7. Weight Changes
Since our gut microbes help to digest the food we eat, they can affect the nutrients we absorb and how we store them as energy in the body. However, the relationship between the gut microbiome and weight changes is intricate, with studies suggesting associations between microbial diversity and weight gain, but causation remains a subject of ongoing research. Obesity has been associated with overall lower gut bacteria diversity. Further, individuals with higher Prevotella to Bacteroides ratio lost more body weight and body fat compared to individuals with low P/B ratio in another study published in Nature.
8. Mental Fog
Mental clarity and cognitive function can manifest as difficulties with focus, memory, and overall cognitive performance and are closely tied to your gut, as mentioned in the above discussion of the gut-brain axis on mood disorders. In patients with dementia who experience cognitive difficulties, certain markers in the gut have been found such as low Bacteroides. A study has also shown that people with brain fog had a higher prevalence of of small intestinal bacterial overgrowth, which is the presence of excessive growth of bacteria in the small intestine.
9. Chronic Fatigue
Chronic fatigue can have a significant impact on your quality of life. You may find yourself feeling exhausted even after a full night's sleep or struggling to get through the day without needing a nap. One large, prospective study found that both Faecalibacterium prausnitzii and Eubacterium rectale were reduced in people experiencing chronic fatigue syndrome (CFS) and that low levels of Faecalibacterium prausnitzii in the gut were associated with worsened fatigue symptoms. Faecalibacterium prausnitzii is a well-known producer of a short chain fatty acid which regulates and dampens inflammatory-related changes in the gut.
10. Frequent Illnesses
Surprisingly, over 70% of your immune cells reside in your gut. Factors that disturb the gut microbiota, for example from diet, stress, or drug treatments, can allow opportunistic pathogens to colonize and grow in the GI tract leading to infection. A stable gut microbiota can prevent these colonizations, as they protect the GI tract by a mechanism called colonization resistance, in which existing microbes compete with invading microbes for resources and prevent their growth. One study reported that certain bacteria like Bacteroides thetaiotaomicron can even trigger the production of antimicrobial peptides that target other intestinal microbes.